Your Cart is Empty



CHEMIX ENERGY
By The Guerilla Chemist
$54.99

Chemix ENERGY is an ultra-premium, natural stimulant-based formula that gives users hours of endless energy. Whether it’s for the gym, work, gaming, or staying up, ENERGY combines 8 ingredients in efficacious and synergistic doses to make sure you are mentally firing on all cylinders.
PRODUCT INFORMATION
-
Each ingredient was chosen for a very specific purpose and dose to ensure that you get the maximum effect every time.
-
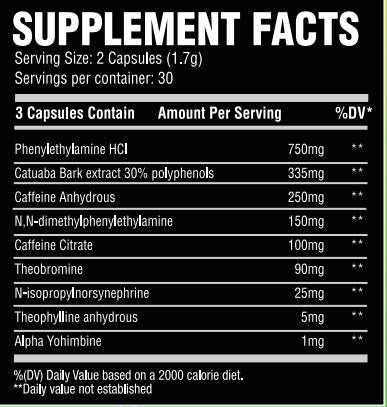
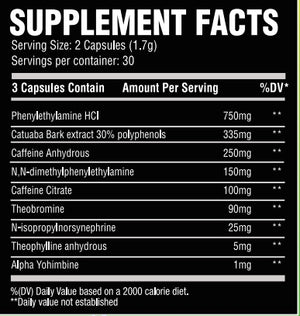
B-Phenylethylamine:Phenylethylamine, or more commonly known as PEA, is the chemical backbone structure for many central nervous stimulants. PEA is found naturally in a variety of botanicals including Leguminosae and even chocolate. PEA as a supplement hits you hard and hits you quickly. That’s why I front loaded ENERGY with 500mg of it for that initial euphoric and stimulant rush.
Catuabua Bark Extract(std to 30% polyphenols): Catuaba (Trichilium catigua) has been used in traditional Brazilian medicine for memory enhancement and libido support. This compound is a superstar ingredient that not only will give you plenty of energy and focus.
Caffeine Anhydrous/Caffeine Citrate: This formula contains a blend of caffeine anhydrous and citrate. Caffeine, a trimethylxanthine, in general is an adenosine antagonist meaning it blocks the sleepiness action of adenosine. This is what causes the alert feeling you get from caffeine. ENERGY has a total of 267mg of caffeine from the 2 sources, not to overwhelm the other stimulants inside this incredible formula.
Hordenine HCL: Hordenine is often mislabeled as a stimulant based on its structure, but it’s actually a mono amine oxidase-B inhibitor. Commonly known as N,N-tyramine, one could see how it can be confused with a PEA derivative. Most companies use hordenine completely incorrectly or at too low a dose to be effective. Not me.N,N-dimethylphenylethylamine citrate: One of the strongest PEA derivatives that is legally able to be used in supplements, N,N-dimethylphenylethylamine citrate has the PEA backbone but with one crucial difference: it contains gemini methyl groups in the Nitrogen atom of the amine group. This allows it to last longer in the body. This will have a similar effect as PEA, but should last longer and hit slightly harder!
N-isopropylnorsynephrine:A newer stimulant to hit the market that is based-ff the ever popular synephrine stimulant, N-isoprpylnorsynephrine has an isopropyl group attached to the Nitrogen amine in order to make its effects last even longer. Synephrine is already a powerful stimulant. N-isopropylnorsynephrine also contains this hydroxyl group and has been shown to burn fat and stimulate your Beta-2 adreno receptors to the legal limit.
Alpha Yohimbine: Last, but certainly not least, I added a very small amount(1mg) of alpha yohimbine. If you recall, alpha yohimbine binds to different subunits of the yohimbine receptor so that you do NOT get the jitters or anxiety that normal yohimbine can elicit. TRUST ME, I HATE regular yohimbine. But, I have found that a very small amount of alpha yohimbine can be very beneficial. Prolonging the activity of norepinephrine can allow you to train harder or just have more energy throughout the day. -
As a dietary supplemment, take one full serving (2 Capsules) with 8-10oz of water. To assess tolerance, one may wish to start with 1 capsule for a few days before progressing to a full serving.

Reviews

ENERGY
Good stuff and not strong. I would recommend this product.

LIFE SAVER
These have literally been a life saver in prep, pure energy, quick shipping high quality for a reasonable price highly recommend. Definitely gonna be ordering more

NO CRASH
Great energy with no crash. I like that you can take a half dose with the serving being two capsules.
Nutritional Facts